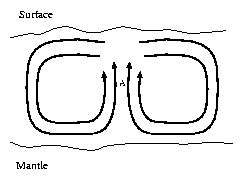
One explanation for this difference in concentration is based on large-scale water flows. Water, present in the pores of the rock, is heated at the lower boundary of the crust, near the mantle, and the change in temperature reduces the density of the water. Beacuse of the lower density this water tends to rise, displacing the water above it. At the surface of the crust the opposite happens: the water cools down and sinks into the crust. Thus large circular flows of water are created, called Bénard convection rolls.
 |
In the upwardly moving sections of these rolls, indicated by the letter A, the water is being cooled down by its surroundings; in the downward moving sections it's being heated. For most minerals, solubility depends on the temperature, and a decrease in temperature means a decrease in solubility at the same time. So the water that has gathered both heat and dissolved minerals near the mantle, will have to release some of the minerals on its way up.
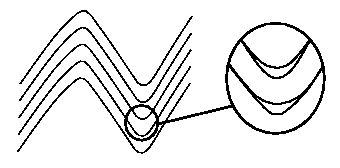
In comes the folded rock. Layers of rock that fold into a hinge need to accomodate large deformations in the hinge zone. This results in voids between the layers, as in the figure below (see the section on Chevron Folding for more about the voids). These voids, and especially the fact that the water pressure is lower in the voids than outside of them, acts as triggers for the deposition of minerals. Over millions of years this process is capable of gathering large quantities of tracer minerals from large expanses of rock and depositing them at very concentrated sites. This may be a mechanism that is responsible for the veins.
 |
To summarize, when searching for veins of valuable minerals, it might be wise to look for highly folded rock, and this is the reason why the mining industry is very interested in the theory of geological folding.